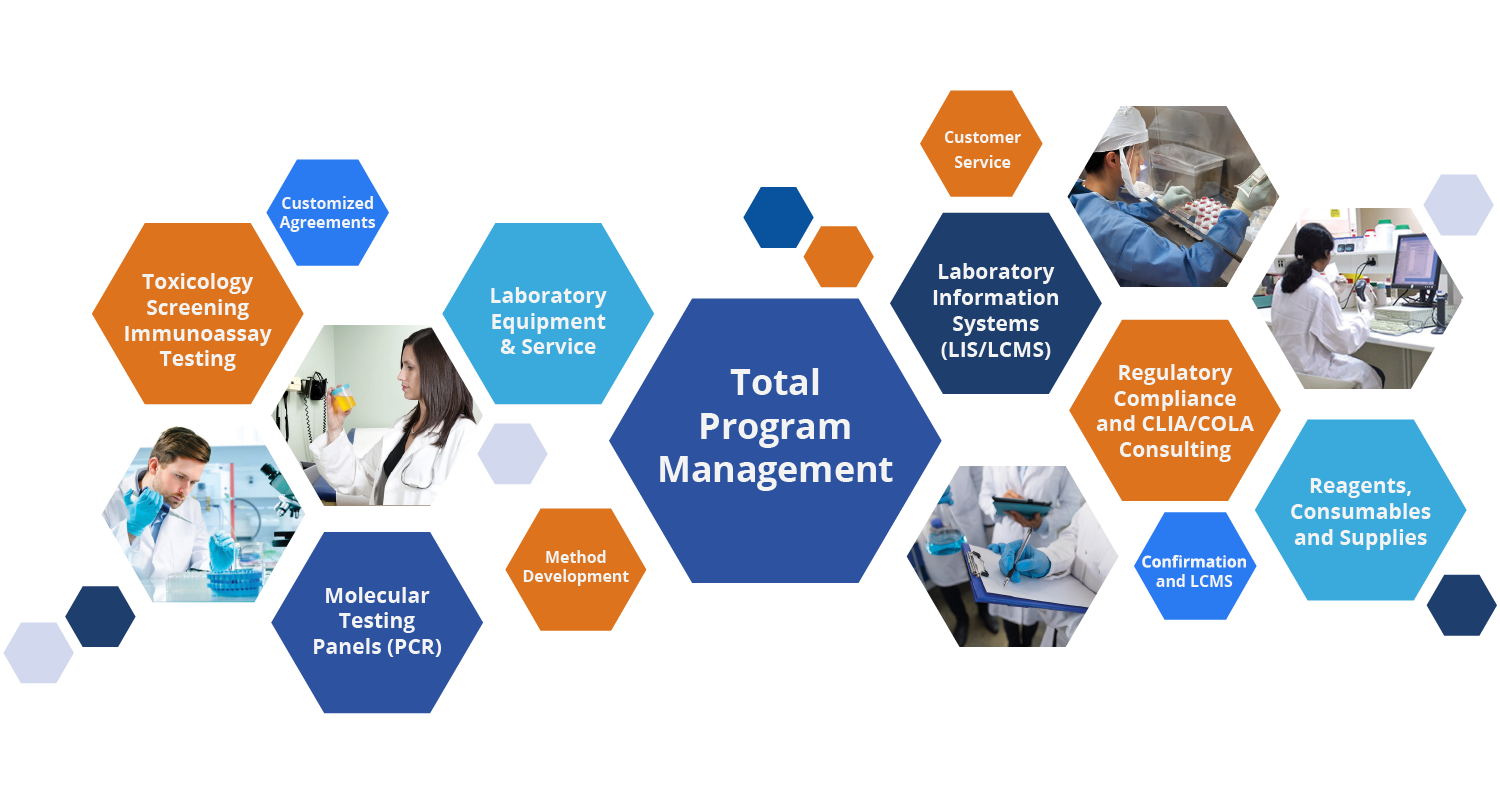
DTPM is a leading provider of turnkey laboratory solutions, currently serving customers in 47 states and growing. Trusted since 1993 to provide a comprehensive array of diagnostic lab testing equipment, supplies, and services. DTPM is your total solution provider.
National in scope. Local in service.

Physician Offices

A physician’s primary focus is to provide excellent patient care. DTPM supports you by providing knowledge, direction, and customized agreements to meet the unique demands of your toxicology or molecular lab.
Reference Labs

Success in a reference lab setting means handling high volume routine and specialty testing, while maintaining accuracy and reliability that healthcare professionals can depend on.
Recovery Courts

Recovery courts have been hailed as one of the nation’s most effective methods for intervention and treatment in the judicial setting. Recovery courts hold offenders accountable with regular and random drug testing.
Treatment Centers

Scheduled drug testing is a key component of monitoring recovery in a drug rehabilitation setting. DTPM assists labs that are affiliated with treatment centers to perform reliable, secure testing.
Our Mission
DTPM’s mission is to support your molecular or toxicology testing facility from start to finish. We specialize in laboratory setup, staffing, procedures, ongoing supply and management, productivity, cost-efficiency, and regulatory compliance.
You own the lab – We make it a success.
DTPM Online Store
We are a distributor of one of the largest selections of Reagents available online. Visit our store to shop for chemicals, lab supplies, equipment and more.